 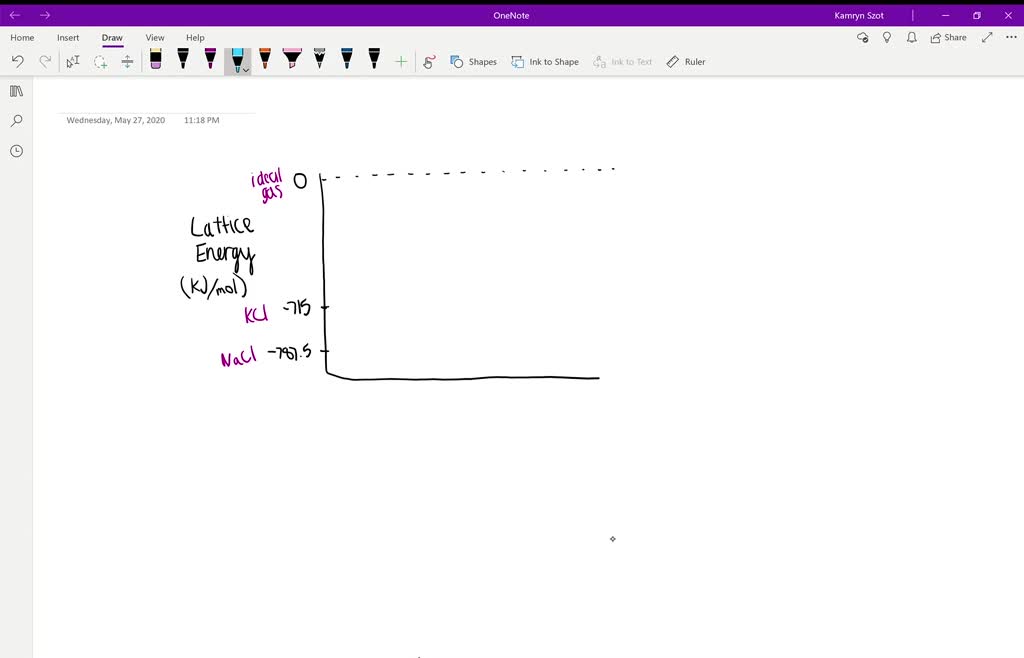
They have the thermodynamic data of Group I and Group II halides in Table 1. This definition causes the value for the lattice energy to always be positive, since this will always be an endothermic reaction. In one definition, the lattice energy is the energy required to break apart an ionic solid and convert its component atoms into gaseous ions. urgently c) Explain why the lattice energy of Magnesium Fluoride is more exothermic that that of calcium chloride. 
Faraday Soc., 1958,54, 34-39, is relevant the OP's question with actual experimental data. Lattice Energy is a type of potential energy that may be defined in two ways. A paper titled The solubilities of some inorganic halides, Trans. So may even ask, which magnesium chloride is being discussed?Īs a result, many chemists rely on macroscopic measurements such as thermodynamic properties but such properties are quiet about microscopic details and "whies". Take anhydrous $\ce$, and add it to water, the reaction is not exothermic in water, and water's pH does not change. Tools such as ChemDraw and SciFinder can provide estimates, but there is a significant margin of error in those solubility numbers and the results may be incorrect. Lattice Energy The energy associated with forming an ionic crystal from atoms Why does magnesium chloride (MgCl 2) have a larger lattice energy than calcium chloride (CaCl 2) Magnesium is in period 3 Calcium is in period 4 Cl ions get closer to the center of positive charge (core) in MgCl 2 than in CaCl 2 Mg2 cations have smaller radius. The size of the lattice energy is connected to many other physical. It is a measure of the cohesive forces that bind ionic solids. The same goes for melting and boiling points. In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. While it may be tempting to come up with explanations that allow us to pass exams or understand concepts on a surface level, the truth is that our current understanding of chemistry is not advanced enough to accurately predict the solubility of compounds in water or other solvents. Although it is very tempting to rationalize every laboratory observation, there are no definite answers to questions about why certain chemical phenomena occur. So factoring 254.66/2 into my equation below i ended up with a lattice energy of -863.33.There are no answers to such why questions, in case anyone is looking for an "answer". I keep finding all sorts of different enthalpy of vaporizations for bromine.free energy of solution of, 19 : 11103 CaCl2 - AgCl, phase diagrams. Use the data given to calculate an overall energy change (in kilojoules per mole) for the formation of from its elements.Įa for F = -328 E11 forCs = 375.7 E12 for Cs = 2422 Heat of sublimation for Cs = 713 CALCIUM COMPLEXES 16 : 23703 binding energy of gaseous, 16 : 28964 chloride. The estimated lattice energy for is CsF2 2347kj/mol.For calcium the first ionization energy is 589.5kJ/mol and the second ionization energy is 1146kJ/mol. Calculate the lattice energy in kJ/mol for CaCl2 from the following information: Energy needed to vaporize one mole of Ca(s) is 192kJ.the difference between the enthalpy of the the difference between the activation energy and reaction enthalpy b. 
A reaction is always non-spontaneous whenĪ.Assume that the energy input in this case is chemical potential energy and the useful The legacy Materials Project website remains available. Consider this energy transformation diagram. Welcome to the new Materials Project website Read our release announcement here. We know that the law of conservation of energy states that energy can not be created or destroyed.The enthalpy of formation of caesium chloride is -44.28kjmol-1 and enthalpy of sublimation of caesium is 77.66kjmol-1.With other data from other sources calculate the lattice energy of CsCl(s)?.What similarities do nuclear energy and chemical energy share? Choose all that apply.Ī. Ionization energy of Na(g): 495 kJ/mol Electron affinity of O2 for 2e: 603 kJ/mol Energy to vaporize Na(s): 109 kJ/mol O2(g) bond energy: 499 kJ/mol Energy change for the reaction calculate the lattice energy of sodium oxide (Na2O) from the following data:. 
Calculate the lattice energy of potassium oxide from the following data:Įnthalpy of sublimation of potassium: 89.24 kJ/mol Bond energy of oxygen: 498 kJ/mol First ionization energy of potassium: 419 kJ/mol 1st electron affinity of oxygen: -141 kJ/mol. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
